-
Larval Food Host
-
Documented natural hostplants are Carex species including
C. appressa (tall sedge), C. fascicularis,
C. gaudichaudiana (Cyperaceae). In captivity, immature larvae prefer
to eat wetland grasses (Poaceae) including
*Ehrharta erecta (panic veldt grass),
Microlaena stipoides (weeping rice-grass) and
*Poa pratensis (Kentucky blue-grass).
However, even though the larvae will eat grasses, limited observations suggest
a diet solely of grass is ultimately toxic to the larvae, just as some grasses
may be less toxic than others. The presence of Carex appressa may be a
necessary obligatory stimulus for egg laying even though the larvae may not
always eat the plant.
-
Eggs
-
Small, pale greenish yellow, subspherical, flattened basally, shiny but with
numerous (35-40) indistinct, very fine, vertical ridges in the upper portion.
The lower portion is very finely dimpled with irregular facets. The top of
the egg is free of ridges and is very finely dimpled with five or six sided
facets. The small, circular micropylar area at the extreme top of the egg is
smooth with a raised edge. Eggs are usually laid singly, but sometimes also
in small groups of two to five, usually on the undersides of the hostplant
leaves, but also sometimes on dead leaf debris within the hostplant. The eggs
have poor adhesive properties and tend to eventually drop off. Some females
may just drop the eggs loose down into the base of the hostplants, which
would be detrimental to the eggs if the plants have water around their bases.
Eggs dropped intentionly by the females may be intended for multiple hostplants.
Larval development within the egg commences immediately after being laid, and
larvae emerge after about 9-11 days in late summer. The eggs require moisture
and humidity for development, without which the eggs will often dessicate.
This may be the reason the eggs have loose adhesive properties, as the basal
parts of the hostplant clump has much better humidity. When ready to emerge,
the larva incompletely cuts out a circular area at the top of the egg, leaving
a small uncut portion, and then pushes the top out like a hinged lid. The egg
shell is then usually eaten by the larva.
-
Larvae
-
The first instar larvae are about 3 mm long, cylindrical shaped, initially pale
greenish yellow, but after eating the green leaves of the hostplants the larvae
acquire a green colour, with darker dorsal and subdorsal lines. The head is shiny,
large, round and dimpled, with some short white setae. Initially the head is brown
to dark brown but it gradually becomes darker as the larva grows and is black by
the end of the first instar. There are scattered short, black simple setae along
the body that are not knobbed at their ends. The posterior end of the larva is not
divided in the first instar.
The second instar is green with longitudinal yellowish dorsal, subdorsal and lateral
lines, and black lateral spiracles. There are some pale, simple, secondary setae,
that are bent and pointed, and the posterior end is weakly divided. The head is round,
with a very fine granular surface, with some very short bristly pale setae. It is dark
brown coloured approaching black in some specimens, with the odd white dot, and there
are a prominent dorsal pair of white triangular marks. The third instar is similar to
the second instar, except there is dark sublateral line along its posterior part.
The head is flattened on top, brown with pale lateral dorsal and lateral markings,
and the posterior end is noticeably divided and pink or brown coloured.
The fourth instar is either green, greenish brown or pinkish brown, with pronounced
darker dorsal and subdorsal longitudinal lines, the head is brownish green or brown
with paler markings.
The mature fifth instar larvae are cylindrical shaped, tapering anteriorly and
posteriorly, slightly flanged laterally, about 25 mm long. The body is without
long hairs, but bears numerous short pointed secondary setae imparting a rough
scabrous appearance. The larvae occur as various colour polymorphs, being either
green, greenish brown, or brown, with a darker dorsal line and finer dark subdorsal
lines, and a blackish lateral line. The dorsal areas are usually paler than the
lateral areas. The head is large, dark reddish brown coloured with paler markings,
very finely rugose, and slightly concave dorsally with a pair of vestigial horns.
The rear end has a short fork.
The immature larvae nibble the leaf edges of the hostplants, but later instars devour
entire leaves. Larvae generally hide around the base of the hostplant during the
day, hiding within the leaves or within debris caught within the plant, coming out at
night to feed. The latter larvae emerge soon after sunset to climb the hostplant,
and move very slowly. If disturbed while feeding, the larvae may release their hold
on the grass and drop to the ground, remaining rigid for some time in whatever position
they were in at the time of the disturbance. This can be terminally dangerous for
the larvae as during winter the hostplants are often standing in shallow water. If
the hostplants have a few dead leaves, then both green and brown larvae are extremely
well camouflaged on the hostplant and very difficult to detect. The larvae pass quickly
through the first three instars during late summer and autumn, then grow slowly through
winter as the fourth instar. Pupation takes place at the end of the fifth instar in
late spring and early summer.
-
Pupae
-
Short, stout, nearly smooth, about 13 mm long, generally rounded anteriorly and
posteriorly, although the head is wedge shaped anteriorly and slightly concave
without horns, there is an indistinct thoracic keel, and the pupa is slightly
ridged laterally at the thorax-wing junction, the wing joints are protuberant,
and the cremaster is short and spinose. Green coloured, with pale yellowish wings,
cryptically marked with black spots, especially on the head, wings and lateral ridge,
and there is a series of subdorsal black spots on the abdominal segments edged
posteriorly with white, and the dorsal keel is outlined with white. The pupa is
suspended head downwards by the cremaster, and pupation usually takes place within
the congested lower parts of the hostplant, but sometimes on adjacent plants or
objects. The pupal duration is variable, being generally longer in the cooler
months or areas, and varies from 25-38 days.
-
Flight Period in South Australia
-
Only one brood a year, with a normally short flight period from mid December
to early February, although there are also single records for mid November
and early March. Like most single brooded satyrid butterflies, the males start
flying earlier in the season than the females.

-
Distribution
-
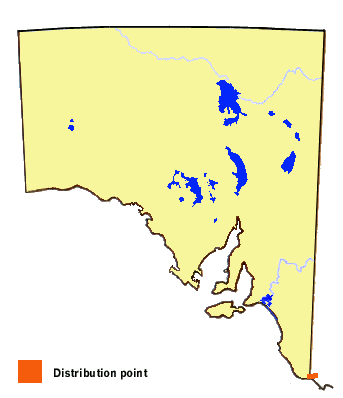
In South Australia, this butterfly is known to occur only in the extreme southeast
corner of the Lower Southeast Region of the state. Historically, its distribution
may have been more extensive before the clearance and degradation of its Carex
habitat. Carex is actually widespread in its distribution in South Australia,
but the early stages of the butterfly require moist, cool temperate conditions for
survival, being limited to areas with annual rainfall in excess of 600 mm. The local
subspecies of the butterfly also occurs in the adjacent parts of western Victoria,
in the Carex bogs and swamps associated with the pristine portions of the
Glenelg River drainage basin. Elsewhere on the Australian mainland,the butterfly
occurs as the nominate subspecies and exists throughout the cooler, damp areas of
the Great Dividing Range in Victoria, and extending through the ACT as disjunct
elevated populations to northern NSW. Three other subspecies occur in Tasmania.
The species is morphologically variable, and in the eastern state localities the
disjunct butterfly populations can have different wing-pattern morphologies.
-
Habitat
-
The butterfly occurs in Carex wetlands that are fed either by permanent freshwater
springs, or in low lying areas having permanently high water tables, or associated with
the Glenelg River and its tributaries. The adults cannot tolerate very hot open areas,
and need shade during hot days. It often flies with the moth
Chrysolarentia
conifasciata
,
which is recognised as requiring sub-alpine habitat! In South Australia, this race is
therefore essentially restricted to a combination of Carex, Gahnia clarkei
and reed wetlands with peripheral Melaleuca and tea-trees (Leptospermum lanigerum),
a habitat that provides guaranteed moisture and shelter. The presence of Carex
provides enough open-ness for the growth of grasses, the likely primary hostplant of
the butterfly. This habitat is also reqired by other rare or threatened butterfly species
in the region, particularly satyrs, such as
Hesperilla idothea,
Oreixenica kershawi,
Oreixenica lathoniella and
Tisiphone abeona.
-
Conservation Status in South Australia
-
The local subspecies wilsoni is Endangered
and forms part of the Butterflies on the Brink group (Geyle et. al. 2021)
which have the potential to become extinct in the next 20 years.
A small colony of the butterfly was observed during the 2004-2005 flight season
(Haywood & Natt, 2006), prior to this it had not been
observed since 1976.
This colony is maintaining a precarious presence in South Australia
where only one small colony is still known to exist.
It was last documented in western Victoria in 1980,
however encouragingly observations have occurred more recently in this region
Other subspecies are locally common in the eastern states
and Tasmania where its preferred habitat is more extensive.
References
Geyle, H. M., Braby, M. F., Andren, M., Beaver, E. P., Bell, P., Byrne, C.,
Castles, M., Douglas, F., Glatz, R. V., Haywood, B., Hendry, P.,
Kitching, R.L., Lambkin, T. A., Meyer, C. E., Moore, M. D., Moss, J. T. Nally, S.,
New, T. R., Palmer, C. M., Petrie, E., Potter-Craven, J., Richards, K., Sanderson, C.,
Stolarski, A., Taylor, G. S., Williams, M.R., Woinarski, J. C. Z., and
Garnett, S. T. (2021). Butterflies on the brink: identifying the
Australian butterflies (Lepidoptera) most at risk of extinction.
Click here for Austral Entomology article.
Haywood, B. T. and Natt, V. (2006). First confirmed observation of
Heteronympha cordace wilsoni Burns (Lepidoptera: Nymphalidae: Satyrinae)
in South Australia. Australian Entomologist, 33 (1), pp5-7.
-
Threats
-
In the southeast of South Australia, nearly all the Carex bogs and swamps
have long ago been cleared, drained and degraded. Carex swamps still occur
in the adjacent parts of Victoria, but they have become severely fragmented, and
a terminal degradation of any H. cordace population in any of these swamps
by either burning, poison spraying, clearing or stock overgrazing can no longer
be repopulated by normal dispersal of the butterfly.
The mouth of the Glenelg River has periodically been allowed to silt up, causing the
water level of the back reaches of the river to become higher and causing the
Carex habitat along the edges of the river to become flooded, to the detriment
of the early stages of the butterfly. The Glenelg River is in a miserable condition,
due to past farming and urban practices that have allowed the banks and adjacent areas
of the river and its tributaries to become severely degraded. Many Carex swamps
are adjacent to timber plantations and are therefore subject to poisonous spray drift
from the aerial applications of insecticides and herbicides used to protect the
plantations during the early stages of their growth. Historically these swamps were
also used to graze stock and usually such areas were drained and periodically burnt
off to promote succulent vegetal regrowth for the stock, all to the detriment of
the butterfly.
Small fragmented areas of Carex can also be colonised by the Spiny Spider
(Jewel Spider) (Austracantha minax). These small spiders live together in
communal groups that can sometimes be very large, having the ability to construct
overlapping webs that can completely enshroud the tops of the Carex.
Without animal activity, either stock or kangaroo, these webs can create a terminal
death-trap for a H. cordace population should it occur or attempt to recolonise.
-
Conservation Strategy
-
Further more determined surveys for this butterfly in southwest Victoria (where the
remnant Carex swamps are more extensive) are urgently required to determine
if the butterfly still exists in that area. A conservation strategy is required
between South Australian and Victorian Threatened Species authorities to protect
the probable last known remaining population area of the butterfly, and to
instigate a repopulation of the butterfly into known secure areas of habitat.
Such areas need to be secured from future degradational impacts. Dedicated areas
revegetated to the Carex hostplant may be required to secure this butterfly.
The butterfly is adaptable to small areas of habitat. The control of mosquitoes in
remaining broad-acre wetlands through the use of broad spectrum insecticides needs
to be judicially controlled, but preferably should cease, as it can be a major cause
of fauna destruction.
Interestingly, this butterfly has recently made an amazing recovery, from the belief
that it was likely extinct, yet during 2005 it suddenly reappeared at its old fragmented
haunts in both western Victoria and southeast South Australia, although its numbers during
2007-2008 have again severely declined!






