-
Larval Food Host
-
Hostplants have not yet been determined in South Australia. In the eastern
states the larvae primarily utilise common grasses. Those hostplants that
also occur in South Australia include *Ehrharta erecta (panic veldt grass),
Microlaena stipoides (weeping rice-grass),
Poa spp incl. P. tenera (slender tussock grass) (Poaceae);
also less frequently on a fine leaved Lomandra sp (Asparagaceae)
and Gahnia sp (Cyperaceae).
In captivity in South Australia, larvae prefer Microlaena stipoides,
but will also develop on *Brachypodium distachyon (false brome) and
sometimes on *Ehrharta erecta. Young larvae will eat the soft new
growth of *Pennisetum clandestinum (kikuyu), but ultimately this
grass is toxic to the larvae. However, mature larvae can tolerate kikuyu
for a short period. Surprisingly, considering how common the adults are
in the wild, the larvae are extremely fussy eaters in captivity, requiring
exacting conditions and hostplant, such as high humidity and the hostplant
to remain in a green condition.
-
Eggs
-
Small, initially white or pale yellow, hemi-spherical (domal) with well developed
vertical ribs numbering from 12-14, and additional finer cross latticing around
the base. Usually laid singly on the hostplant or on ground vegetal-debris next
to the hostplant, also sometimes laid in pairs or in short rows. The female
sometimes leaves abdominal hairs on the eggs, presumably for camouflage.If the
egg is fertile, then after a couple of days it acquires a purplish red dorsal spot
and a jagged purplish-red lateral band. Larval development within the egg commences
immediately after being laid, and the larva is ready to emerge in about 13 days.
However, the larva will not emerge until it is wetted by autumn rain, usually
combined with a seasonal drop in air temperatures. They will sometimes eat out the
top of the egg, and then just sit inside the egg with their black head blocking the
opening, waiting for the rain to come. In captivity the larvae can remain alive
inside the egg in a diapause (torpid) condition until early June. The egg shell is
eaten by the larva after it emerges. The larva then moves off to find suitable
hostplant, testing all likely grass by taking a small nibble out of the leaf edge.
When it finds a suitable hostplant, it will move to near the end of the leaf and
make a small tubular shelter within which to hide, usually by foldingthe leaf-end
over, but sometimes by rolling the side edges of the leaf, and binding the edges
together with silk. In the case of the latter shelters the opening facing towards
the base of the hostplant is eventually sealed with a plug of silk, leaving a single
opening facing towards the end of the leaf.
-
Larvae
-
The first instar is long cylindrical and initially yellowor yellowish white,
with a large black circular head that is smooth and shiny, the upper front
part weakly grooved longitudinally. There are a few short hairs on both the
body and head, that are slightly longer at the posterior end. The neck
(prothoracic plate) is dark wine red coloured. After eating the hostplant,
the larvae gradually become greenish. The second instar is similar to the
late first instar but has a distinct, broad white longitudinal dorsal band
split by a central narrow green dorsal line, and there is a further narrow
white subdorsal line. The posterior end is brownish. The third instar is
similar to the second instar but differs by having a brown, rugose head and
the prothoracic plate is brown. New shelters are periodically constructed to
fit the growing larvae. The early instar larvae feed at night by eating the
leaf edges below the shelter, and leaving the central mid-rib of the leaf.
Eventually the shelter is suspended by only the wiry remains of the mid-rib vein.
The fourth instar is similar to the final instar. At this stage the larvae have
usually outgrown the width of the hostplant leaves, and if the leaves are not
wide enough (as is the case of most of the host grasses) then the larvae will
cease to make shelters from the leaves of the host grass and start to use loose,
rolled leaf and bark debris as shelters, either caught up within the host grass
or lying loose on the ground around the base of the grass. Once selected the leaf
or bark shelter is lined with white silk, with one end sealed off with strong
strands of silk, and the shelter is secured to an adjacent object also with strong
strands of silk. Enough silk is used so the shelter retains its shape during dry
and wet spells of weather. The shelters are not always occupied by the same larva
and may be shared around with other larvae, presumably based on the principle of
'first in first served' after a hard nights feeding. Larval feeding is during the
night, and there is slow growth during the winter months but there is flourishing
eating activity during the warmer spring months and most larvae reach the final
fifth instar stage by mid spring.
The fifth and final instar is fat and humped, typical of the Trapezitini Tribe, about
18-20 mm long, initially green with an indistinct pale, broad dorsal band and a darker
central dorsal line,and a distinct narrow white subdorsal line. The lateral parts are
pale while the posterior end is brownish. The neck area (prothoracic plate) is similar
to the body colour. The head is large, oval shaped from the front, rugose, centrally
grooved longitudinally, strongly notched at the top, mostly dark brown-black coloured
but with a diffuse pale brown line either side of the central groove in the upper part,
a large pale brown inverted V mark in the front lower part, some additional diffuse pale
brown markings in the large frontal areas either side of the central groove but which
do not reach the basal part of the head. The head is covered with very short forward
directed, curved setae. The head pattern markings are distinctive for the species and
differ from S. flammeata by being
generally much darker. The lateral edge of the larva is strongly flanged.
The body is without long hairs, but is covered with numerous white, elongated
wine-glass shaped secondary setae set on
simple smooth, raised bases, that are further set in a small white circular area that
impart a white dotted appearance to the larvae. These setae impart a rough scabrous
appearance. When fully grown and during the pre-pupa stage the larva turns brownish.
The final instar larvae continue to eat and grow strongly into early December but then
slow or cease to eat and become quiescent. By the latter half of December pupation starts
to occur among the larvae and these larvae then pupate at various times and some may not
pupate until late summer or early autumn. These later larvae may periodically feed to keep
up their energy levels. The larvae select shelters having a very tight fit.
-
Pupae
-
Short cylindrical, typical of the Trapezitini Tribe, about 12-15 mm long,
brown, but darker brown anteriorly and posteriorly, and (like a close
relative Anisynta cynone) is copiously
covered all over with a white water repellent powdery bloom (probably
also with anti fungal-bactericide properties) that is very easily dislodged
by any wriggly movements by the live pupa, producing an irregular pattern
of brown under-markings. The thorax of the pupa is humped dorsally. The head
is weakly flattened anteriorly, with the head cap (operculum) being dark
brown coloured. The front (anterior) part of the operculum, and again laterally
behind the eyes, is weakly sclerotised (finely rugose) with short, fine simple
brown hairs. The central front part is weakly furrowed longitudinally. There are
three pairs of black spots without bloom on the head area, two of the pairs occur
in-line dorsally across the junction of the head with the thorax, a third smaller
pair occurs ventro-anteriorly on the front of the head. A large fourth pair of head
spots of dark brown colour, sometimes occurs anteriorly between the eyes. There are
sparse, forward directed, short simple setae (hairs) over most of the pupa (excepting
the wing areas), that become bristly, very common and posteriorly directed on the
abdomen. The posterior end tapers to a large fan-shaped cremaster, which is dark
brown coloured, flat, expanding similar to the paired flukes of a whale or dugong
tail. The dorsal area of the cremaster is centrally furrowed and has a series of very
fine elongate ridges or striations, and long cremaster hooks emanate from the ventral
posterior edge. On each side of the cremaster is a short thorn-like smooth spine.
The shape of the cremaster is diagnostic for the species.
Pupation occurs in the final larval shelter after one end of the shelter is sealed with
a white web of silk. The shelter opening at the head-end of the pre-pupa is usually
left open, although sometimes it is partially blocked by some strong strands of silk,
probably to prevent other larvae from trying to enter the shelter. The pupa is secured
within the silk lined shelter by the very strong, cremaster hooks. In captivity in
Adelaide the pupal duration varies from 15-20 days during early summer, but is likely
to be longer in the cooler Southeast Region and the longest period reported is 25 days.
The empty pupal case remains inside the shelter after the adult skipper emerges, and is
brown coloured.
-
Flight Period in South Australia
-
The main flight is from early January to mid March, with the odd skipper occurring
into early April. Only one brood a year.

-
Distribution
-
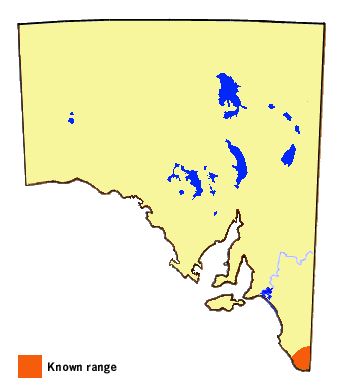
In South Australia it is presently confined to the Lower Southeast Region in areas
with annual rainfall above about 600 mm. The skipper is also found in the higher
rainfall areas along the coast and Great Dividing Range in Victoria and extending
to as far north as southern Queensland.
-
Habitat
-
The skipper usually occurs in moist grasslands and open forest with a grassy
understorey, growing in cool temperate areas. The early stages of the skipper
require continuous humid conditions.
-
Conservation Status in South Australia
-
Local in its distribution. Sometimes locally common during peak periods of emergence.
-
Threats
-
The main threat is from toxic spray drift due to the farming and forestry
practice of aerial application of insecticidal sprays.
-
Conservation Strategy
-
Suitable habitat is reasonably common. Applications of toxic sprays,
particularly by aerial means, in farming and forestry areas should
be judiciously applied.










