-
Larval Food Host
-
Native and introduced citrus type Rutaceae including garden *Citrus species
(grape-fruit, lemon, lime, mandarin, orange, etc), Citrus (Eremocitrus) glauca
(desert lime), *Citrus (Fortunella) species (kumquat), Geijera parviflora
(wilga), *Citrus (Poncirus) trifoliata (trifoliate orange, wild orange) (Rutaceae).
The female prefers to lay eggs on the large-leaved, introduced types of citrus, and does not normally
go out of its way to lay on kumquat or desert lime. Larvae normally eat the leaves and soft new growth
of the hostplant, but sometimes the flowers and buds are eaten. It has been observed that during its life
period a single larva will eat the equivalent of about two fully developed leaves of a grape-fruit tree,
which would translate to about three leaves of a lemon or orange tree. The female is not known to use
Geijera in South Australia.
-
Eggs
-
Large, pale yellow, nearly spherical, basally flattened, smooth. Near larval emergence the eggs
change to an orange colour. Laid singly on the hostplant. The female will only lay eggs on
the new growing tips or soft new leaf growth of the hostplant. On the larger leaves
they are laid at the leaf edge, both above and beneath. Larval development within
the egg commences immediately after being laid, and the eggs normally hatch in about 3-4
days but sometimes within one day if the female has been holding on to them for a long
period while they are fertilised. Many of the eggs are also laid sterile. The
egg shell is eaten by the larva after its emergence. Females will often patrol a set
urban area containing its citrus hostplants, returning at set times during the day, to
check for new growth. The eggs are quickly targeted by various small predators that
suck out the contents of the egg and are also commonly parasitised by tiny wasps.
-
Larvae
-
The first instar is dark coloured, with two subdorsal rows of short, bristly, fleshy spines.
The anterior, middle and posterior parts have broad transverse yellowish bands, giving the
larva a 'bird dropping' camouflage pattern typical for the swallowtail group of butterflies.
The first three larval instars retain this pattern. Head black, smooth, shining, with
some short hairs, later instars have an inverted white V mark on the front which becomes
most prominent in the final instar. The fleshy spines become less bristly in later
instars, and branching in the final instar is virtually absent. The immature larva eats
the egg shell after emergence, before proceeding to eat new leaf shoots. Later
instars devour fully developed new leaves, but not normally those leaves that have matured
and hardened. In warm areas the larvae pass through five instars, although those
occurring in cold areas may require six instars to fully develop to the pupation stage.
Mature larvae about 35 mm long, cylindrical shaped, black coloured generally, with two
subdorsal rows of short fleshy black spines (tubercles), and dorsal, sublateral and
lateral rows of large orange/yellow blotches, and numerous scattered small pale blue and
white spots. The amount of yellow is variable, and some larvae can be very yellowish
and pale. The amount of orange is also variable, and in some larvae it is
absent. There are vestigial subdorsal blue 'warning eyes' on the anterior part of
the larva, and also associated with the base of the tubercles, which is typical for the
swallowtail group, although in other species they are usually more obvious. In this
case the small eyes are very similar to those found on the larvae of some members of the
Heraclides group of swallowtails in South America. The head is large, black,
with an inverted white V mark on the front. The mature larva has bold aposematic warning
colours and remains exposed on the hostplant. The colours would appear to be a further bluff
to predators as the larvae are not known to be unpalatable. The mature larvae are
similar to the larvae of other mimic swallowtails found
outside
of Australia. They also sometimes mimic the larvae of the models, and in this case
there is a vague similarity to the larvae of the
Greasy Swallowtail,
which have dark fleshy spines and creamy white patches.
All larvae stages feed exposed on the hostplant. When disturbed, the larvae can
evert a reddish orange coloured, fleshy bifid osmeterium
from behind the head that emits a distinct citrus-like pungent odoriferous secretion,
smelling like rotting oranges on the ground beneath an orange tree. This secretion acts
as a deterrent to both vertebrate (birds, lizards and mice) and invertebrate
(ants, spiders and wasps) predators. It is more effective if the chemical can be deposited
on the predator, hence the larvae will attempt to throw their heads (and osmeterium) either
backwards or sideways if a predator attacks from the rear or side. The secretion is usually
composed of a butyric acid compound having irritant properties. The osmeterium is
present in all stages of the larvae, and is found in all the Papilionidae group of
butterflies. In other species the osmeterium can have a different colour such as
green, blue or black.
-
Pupae
-
Slender, straight, stick like, about 30 mm long, anterior end produced into a short pair
of projections, and the thorax has a prominent, slightly forward directed dorsal projection.
It is usually attached to the stems of the hostplant and nearly always on the other side
of the tree from where the larva was last feeding. The larva will sometimes leave the hostplant
to pupate. The pupa is attached to the stem by a posterior cremaster and a central silken
girdle. It either reclines from the vertical or is suspended horizontal. The colour is dimorphic,
typical for many swallowtails, being either green or grey brown with other variable cryptic
markings. The colour pattern imitates the stem to which the pupa is attached, such that it
looks like a broken extension to the stem. If the stem is green or the surrounding parts are
leafy, then the pupal colour is mostly green, or if the stem is brown without nearby leaves
then the pupa is grey or brown. The pupal duration is variable. It can be as brief as
two weeks in summer, extending to four weeks in autumn, but often those pupae formed in
autumn will not produce adults until the following spring, or even longer with diapause
records of one to two years!
-
Flight Period in South Australia
-
It flies during the warmer months, usually from October to early May.
There are continuous broods over the warmer months, which can be completed in
as little as 5-6 weeks. It normally overwinters as pupae.

-
Distribution
-
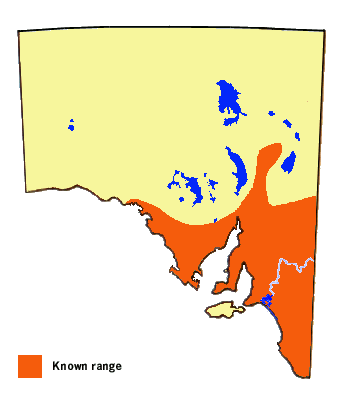
Originally a subtropical eastern states butterfly with vagrant tendencies.
The species has spread to South Australia along the Murray Valley with the development
of irrigated Citrus orchards in that region. It was first documented from South
Australia in the late 1920's, although it was probably active in the Riverland (Murray Valley)
since the inception of major irrigation at the turn of the last century as the butterfly
was known from Mildura in nearby Victoria by 1914. It can now be found in orchards and
most large urban areas in South Australia wherever Citrus trees are cultivated,
and one should not be surprised to see the odd one flying outside of its known range.
However, it is yet to be recorded on Kangaroo Island where likely there is presently
insufficient density of citrus trees, but as the local towns become larger in the near
future the butterfly will probably establish itself. The butterfly has also yet to make it
to Western Australia, but again it is only a matter of time before it is accidentally
introduced to Perth. This butterfly is endemic to Australia. Its occurrence on New Caledonia
is due either to an introduction on citrus trees obtained from Australia or as wind blown vagrants.
-
Habitat
-
An open forest and woodland butterfly in its eastern states habitat. A native hostplant
Citrus (Eremocitrus) glauca (desert lime) occurs in the northern pastoral areas
but is too rare to support viable populations of the butterfly. It is generally considered
to be an urban and orchard butterfly in South Australia.
-
Conservation Status in South Australia
-
A local butterfly, rarely common.
-
Threats
-
It is considered a minor pest in Citrus orchards and is not normally sprayed,
but will succumb when other Citrus pests require spraying.
-
Conservation Strategy
-
Butterfly numbers are not deteriorating in South Australia, so a conservation strategy
is not required. In the urban environment its larvae have the potential to defoliate young
Citrus trees, but this can be managed by hand removal or thinning of the larvae.
They may even be beneficial in some cases by keeping the tree pruned. Larvae numbers are
usually low and natural predators tend to keep numbers in check. Established Citrus
trees are not troubled by the larvae.








