-
Larval Food Host
-
Native and introduced grasses including Rytidosperma species including
R. pilosa (velvet wallaby-grass),
*Brachypodium distachyon (false brome),
*Ehrharta erecta (panic veldt grass),
Poa spp incl. P. crassicaudex (tussock grass),
P. poiformis (coast tussock grass),
*P. pratensis> (Kentucky blue-grass),
P. tenera (slender tussock grass),
Themeda triandra (kangaroo grass) (Poaceae).
In captivity, the larvae will eat many other types of grasses (with variable success)
other than those mentioned above. The larvae feed on the leaves and other soft green
parts of the grass.
-
Eggs
-
Small, pale yellowish white, shining, turbinate to subconical, expanding basally,
base flattened, apex rounded, nearly smooth but with very fine, indistinct dimpling.
The females lay eggs by fluttering close to the ground, then settle on some grass
or stem and proceed to lay. They may attempt to lay eggs on the grass, but as the
eggs have very poor adhesive properties they tend to immediately drop off. Most
females just drop the eggs loose down into the base of the grass. The females have
the potential to lay large quantities of eggs. One female in captivity once laid
more than 300 eggs within a week before she got tired of it, with barely a reduction
in abdomen size. The eggs start to develop as soon as they are laid, and hatch in
about 17-19 days in mid-Autumn. Eggs may develop in 7-10 days if kept warm, while
eggs laid in cold areas may take a month to develop. The presence of moisture will
also sometimes hasten development. When ready to emerge, the larvae incompletely
cut out a circular area at the top of the egg, leaving a small uncut portion, and
then push the top out like a hinged lid. The larvae eat their egg shells on emergence.
-
Larvae
-
Initially, pale greyish yellow, with sparse, long semi-translucent brownish hairs
that have transparent knobs at their tips, and which are set on short, simple
raised protuberant smooth bases. The hairs are slightly longer at the posterior
end of the larva. The head is pale brown, very finely rugose, having short hairs
with only the dorsal hairs being knobbed. The posterior end is not forked in the
first instar. After eating the green leaves of the hostplant, the larvae turn
green and acquire a dark dorsal line edged pale yellow, and pale yellowish
subdorsal and lateral lines, and the head becomes dark brown.
The second and third instars are green. The long, knobbed hairs that are present
on the first instar are lost in later instars, and the rear end becomes forked,
typical for the Satyrinae. The body has a darker dorsal line, a pale subdorsal
and lateral line, and the dorsal area is often paler than the lateral area.
They are covered in short, simple spine-like setae set on white, simple raised
protuberant smooth bases. By about the third instar, the lateral edge of the
larvae becomes noticeably flanged. The head is finely rugose with some short
spine like hairs, rounded, brown, with a darker brown frontal triangular mark
edged pale brown. The forked tail is short,pale yellowish coloured, and is also
covered in setae.
The fourth instars are either pale brown, greenish brown or green, with a dark brown or
black dorsal line and other subdorsal lines, the head is brown, while the forked tail is
pale brown.
The mature fifth instar larvae are cylindrical shaped, 27-32 mm long, being fatter in
the middle, and the lateral edge is slightly flanged. Only brown larvae have been
recorded. There is an unbroken dark brown or black dorsal line, and finer subdorsal
dark lines, and there are other very fine speckled dark markings. The body is without
long hairs, but bears numerous short pointed secondary setae imparting a rough
scabrous appearance. The head is large, rugose and brown with numerous short hairs.
The top of the head is rounded, without horns. The rear end is forked.
The immature larvae nibble the leaf edges of the hostplant, but later instars devour
the entire leaf and also other soft green parts of the grass. Early instar larvae
generally remain exposed on the hostplant, but third and later instars hide around the
base of the hostplant during the day, coming out at night to feed. The latter larvae
emerge soon after sunset to climb the hostplant, and move very slowly. If disturbed
while feeding, the larvae will release their hold on the grass and drop to the ground,
remaining rigid for some time in whatever position they were in at the time of the
disturbance. If the grass has a few dead leaves, then both green and brown larvae
are extremely well camouflaged on the hostplant and very difficult to detect. The
larvae feed slowly through winter and spring, and pupation takes place in mid-summer.
Mature larvae of H. penelope tend to differ from H. merope in that the
dark dorsal line on H. penelope is unbroken, and the top of the head is rounded,
whereas in H. merope the dorsal line is broken and the top of the head is flat.
-
Pupae
-
Pale brown with darker brown or black cryptic markings, 15-17 mm long, short and fat,
with a roughened surface, generally rounded anteriorly and posteriorly, although
the head is wedge shaped anteriorly without horns, there is an indistinct thoracic
keel, and the pupa is slightly ridged laterally at the thorax-wing junction, the
wing joints are protuberant, and the cremaster is short and spinose. Pupation takes
place either at the base of the hostplant in a silk lined cavity formed by the larvae,
or on the ground where the pupation occurs beneath vegetation debris in a shallow
larvae formed cavity in the soil loosely lined with silk. The pupae lie loose inside
the cavity. The pupal duration is about 31-34 days in late summer.
-
Flight Period in South Australia
-
Usually early February to late March, extending into early April during cool seasons.
Only one brood a year. The males start to emerge earlier than the females. The flight
of the females and the hatching of their eggs probably coincides with the drop in
Autumn temperatures and the beginning of the Autumn rains that promote a new flush
of growth in the larval hostplant grasses.

-
Distribution
-
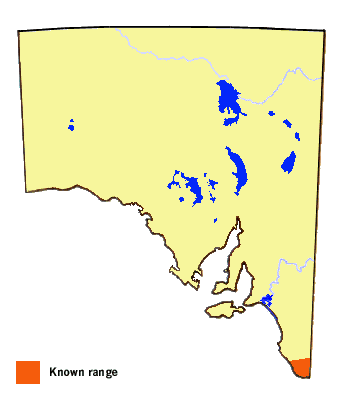
In South Australia, the butterfly only occurs in the Lower Southeast Region,
south of Latitude 37° 30' (approximately a line through Millicent to Nangwarry).
Historically, before the major native vegetation clearance occurred in the region,
the butterfly may have extended further north in line with its distribution in
western Victoria. The local subspecies of the butterfly is also found in similar
habitat areas of Victoria, New South Wales and southern Queensland, and in elevated
areas of the Great Dividing Range in central Queensland. There is some clinal
variation in colour and pattern over its extended mainland range, but all mainland
forms are now grouped under the nominate subspecies. Two other subspecies are found
in Tasmania.
-
Habitat
-
The butterfly occurs in shrubland, woodland and forest, in both open and closed habitat,
which has some grass in the understorey that remains in a green condition all year round.
It can sometimes be found in open coastal Poa poiformis grasslands. It only occurs
in moist habitats receiving more than about 600 mm of annual rainfall. It is not as drought
tolerant as Heteronympha merope, but is more tolerant of dry conditions than the
other members of Heteronympha.
-
Conservation Status in South Australia
-
Local in distribution, sometimes common in its preferred habitat, but otherwise rare.
-
Threats
-
Bushfires, agricultural and forestry expansion are the main threats. State native
forests in the Lower Southeast historically had their understorey burnt off on a
short term rotational basis, which promoted the fire tolerant bracken to take over
the forest understorey, to the exclusion of native grasses and other diverse native
understorey vegetation.
-
Conservation Strategy
-
Its hostplant is common and widespread, so the main strategy would be to keep toxic
agricultural and forestry insecticides and weedicides away from conserved habitat,
particularly those sprays distributed by aerial means.



