-
Larval Food Host
-
Native and introduced grasses including
*Brachypodium distachyon (false brome),
*Ehrharta longiflora (annual veldt grass),
Microlaena stipoides (meadow rice-grass),
*Pennisetum clandestinum (kikuyu),
Poa species including P. clelandii (matted tussock grass),
P. tenera (slender tussock grass),
Themeda triandra (kangaroo grass) (Poaceae).
Early instar larvae feed on the hostplant leaves. The larger final instar larvae
devour all the softer parts of the hostplant including young seed heads.
-
Eggs
-
Small, initially pale yellow or white, turbinate (expanding basally),
the base and apex are flat, moderately thick shelled, with well
developed vertical ribs numbering 24-27, which do not extend onto
the apical flattened area. The top flattened area is not rimmed with
a ridge. Laid singly on dead twig and similar debris that is lying
on the ground adjacent to the hostplant. If the egg is fertile it
becomes purple speckled after a couple of days. Larva development
within the egg usually commences immediately after being laid, and
larvae begin to emerge in about 15 days. The empty egg shell is then
eaten. (In captivity, most of the newly emergent larvae then become
immobile. Some eventually move off to look for the hostplant, while
the remainder stay put and would eventually die unless forcibly moved
onto hostplant). However, some delayed egg development occurs, and
some developed larvae remain inside the egg until wetted by rain.
Some larvae do not emerge until late April, some two months after the
eggs are laid.
-
Larvae
-
Initially pale grey with fine longitudinal purplish brown lines,
with numerous long pale hairs that emanate from black bases.
These hairs are knobbed at the tips. The head is finely rugose,
dark brownish black with some long hairs. In the first instar,
the head is not horned and the rear end is only vestigially forked.
After eating the green leaves of the hostplant, the larvae gradually
turn green and acquire indistinct yellowish lines. The second instar
is green with longitudinal yellow and dark green lines, the head is
green and has a pair of vestigial conical horns, and the rear end has
a short fork typical for the Satyrinae. The horns and forked tail are
pink tipped. Later instars are either green or brown, and with successive
instars, the body hairs are replaced by secondary setae, and the
head acquires increasingly
larger horns.
The mature larvae are long cylindrical shaped, tapering anteriorly and posteriorly,
slightly flanged laterally, about 27-29 mm long, usually green coloured, with narrow
yellow lateral and subdorsal lines, the latter edged with dark green, and there is
a dark green dorsal line. The body is without long hairs, but bears numerous pointed
secondary setae. The head is large,
finely rugose with numerous short hairs, with a well developed pair of flattened
pointed horns, green coloured with the perimeter of the head outlined in maroon which
is further edged yellow posteriorly, and there is sometimes a pair of large dark
maroon spots in the front just above the mouth. The rear end is forked, marked yellow
laterally and the tips are pink. Brown larvae are occasionally seen, in which all the
green colour is replaced by brown, with the dark aspects and yellow and pink markings
remaining the same. These brown larvae presumably develop in response to a hostplant
with many dead leaves.
The immature larvae nibble the leaf edges of the hostplant, but later instars devour
the entire leaf and also other soft green parts of the grass. Early instar larvae
remain exposed on the hostplant, but later instars hide around the base of the
hostplant coming out at night to feed. The latter larvae emerge soon after sunset
to climb the hostplant, and move very slowly. If disturbed while feeding, the larvae
will release their hold on the grass and drop to the ground, remaining rigid for
some time in whatever position they were in, at the time of the disturbance. They
are extremely well camouflaged on the hostplant and very difficult to detect. The
larvae feed throughout winter and pupation takes place in late spring and early summer.
-
Pupae
-
Small, stout, about 13 mm long, with a roughened surface, rounded posteriorly,
wedge shaped anteriorly with a pair of small horns, dorsally keeled, ridged
laterally at the wing junction, and also dorsally across the abdomen. A few
small protuberances ventrally. Usually green coloured, with some darker green
markings, suffused yellow which is best developed ventrally, with six pairs
of raised yellow subdorsal abdominal spots, the dorsal keel and the lateral
and abdominal ridges are outlined with dark brown edged pale yellow, the
yellow outline is strongly developed on the abdominal ridge. There is also
a pair of raised black dots ventrally. As with the larvae, some pupae are
brown coloured, in which the green colouration is replaced by brown. However,
a few days before the butterfly emerges, a green pupa will turn brown as the
butterfly inside the pupal shell develops its natural brown colour. First
the wing cases turn brown, then later the head and thorax, and finally the
abdomen. Brown larvae do not necessarily produce brown pupae. The pupa is
suspended head downwards by a cremaster, either from the hostplant, or from
a log, stick, rock or another plant adjacent to the hostplant. The pupa stage
lasts about three weeks. Near Adelaide it is about 17-19 days during early summer.
-
Flight Period in South Australia
-
Only one brood a year with an extended flight from late November to late March.
The males emerge earlier than the females. The bulk of the males emerge during
late December - early January, while the bulk of the females emerge during late
January - early February. By the end of February the flight is mostly over.

-
Distribution
-
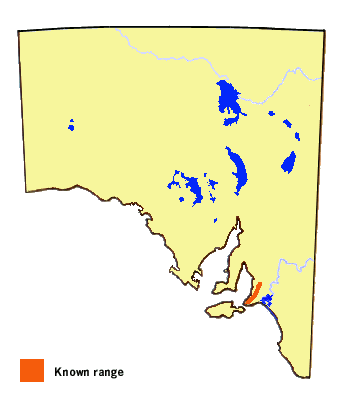
The butterfly occurs only along the South Mount Lofty Ranges in the Adelaide Hills
and along the west side of the Fleurieu Peninsula. Enigmatically, the butterfly
does not occur on Kangaroo Island or in the Lower Southeast of the state where
suitable habitat is present. The butterfly is much more common in the eastern
mainland states of Australia where another subspecies occurs.
-
Habitat
-
The butterfly occurs in cool, damp shady woodland and forest areas receiving
more than about 600 mm of annual rainfall, and having its grass hostplant
in the understorey remaining in a green condition all year round. Its original
habitat (prior to the introduction of invasive grasses from overseas), was
probably the Poa grasslands which require moist conditions.
-
Conservation Status in South Australia
-
Rare.
-
Threats
-
Bushfires, drought, and urban and agricultural expansion are the main threats.
As the habitat of the butterfly is now very limited, introduced woody weeds
such as blackberry, boneseed, bridal creeper, broom and gorse also have the
capacity to be a serious threat by choking off its hostplant and habitat.
-
Conservation Strategy
-
Population numbers do not seem to have deteriorated over the past 30 years,
and it is likely the butterfly is reasonably well protected in the conservation
parks along the Mt Lofty Range. The introduced weeds will have to be monitored
in its conserved habitat, and destroyed where necessary.








