-
Larval Food Host
-
Gahnia clarkei (tall saw-sedge or cutting grass),
G. radula (thatch saw-sedge),
G. sieberiana (red-fruit saw-sedge) (Cyperaceae).
Larvae have only been found on Gahnia clarkei in South Australia. The larvae will
readily transfer onto G. seiberiana in captivity, but this plant is not present
in the Lower Southeast. The larvae feed on the young leaves of the hostplant.
-
Eggs
-
Large, green, subspherical, flattened basally, nearly smooth, shiny but with
some indistinct, very fine, incomplete vertical ridges near the top of the egg.
Eggs are laid singly on the hostplant, usually on the outer young growth of the
hostplant, hidden from sight on the inside of the leaves. Larval development
within the egg commences immediately after being laid, and larvae begin to emerge
in about 10-11 days in late summer. When ready to emerge, the larva incompletely
cuts out a circular area at the top of the egg, leaving a small uncut portion,
and then pushes the top out like a hinged lid. The egg shell is then usually eaten
by the larva. The larvae then become very mobile, looking for the new Gahnia
growth. When they find a place to their liking they will eat a small triangular
piece out of the edge of the leaf, and if to their liking they will then settle down,
hidden within the new growth.
-
Larvae
-
Initially, pale yellowish green, with sparse, short dark hairs that have transparent
knobs at their tips, and which are set on short, simple raised protuberant smooth
bases. The hairs are darker and slightly longer at the posterior end of the larva.
The head is black, very finely rugose but shining, with a few short hairs with only
the dorsal hairs being knobbed, and with a pair of tiny vestigial dorsal horns.
The posterior end of the larva is not forked in the first instar. After eating the
green leaves of the hostplant, the larvae turn green and acquire a dark dorsal line,
and pale yellowish subdorsal and lateral lines.
Subsequent instars are very similar in shape in colour, being long cylindrical and
pale green coloured, with indistinct darker dorsal and subdorsal lines. The dorsal
area of the larvae is usually paler than the lateral area. The long, knobbed hairs
that are present on the first instar are lost in later instars, and the rear end
becomes forked, typical for the Satyrinae. They are covered in short, simple
spine-like setae set on a white, simple raised protuberant smooth base. The head is
green coloured, square shaped, without the vestigial horns, and is finely rugose
with some short spine like hairs. The forked tail is well developed,and is also
covered in setae.
The final instar larvae are long cylindrical shaped, about 42 mm long. Pale green
coloured with darker dorsal, and subdorsal lines. The dorsal area of the larvae is
usually paler than the lateral area. The body is without long hairs, but bears
numerous short pointed secondary setae imparting a rough scabrous appearance. The
head is large, slightly rugose, with numerous short hairs. The top and bottom of the
head are flattened, creating a square shape. The rear end is forked, and tipped
pink.
The immature larvae move to the new growth of the hostplant after eating the egg shell,
and hide within the closely overlapping leaves with their head pointing upwards.
They feed by nibbling the leaf edges of the young growth. Later instars move to the
slightly older leaves where there is more room to manoeuvre. Early instars have a
characteristic eating style of eating out triangular shapes in the leaf edge, but older
instars will eat out large angular chunks from the leaf edge, often completely chewing off
the end of the leaf. The earliest instars will feed at various times during the day
or night while they are well hidden amongst the young leaves, but older instars remain
hidden on the hostplant during the day, coming out at night to feed. They hide
during the day at the inside base of the long leaf they are feeding on, well hidden within
the confined space between two leaves, usually with their head directed downwards.
Their green colour usually provides good camouflage.
-
Pupae
-
Elongate, slender, nearly smooth, about 18-19 mm long, tapering posteriorly,
wedge shaped anteriorly with a pair of widely spaced short horns, there is
a thoracic keel, the pupa is ridged laterally at the thorax-wing junction,
and the cremaster is long and spinose. The pupae are green coloured, with
the lateral ridge marked yellow. The pupae are suspended head downwards
by the cremaster, and pupation usually takes place within the congested lower
parts of the hostplant, but sometimes on adjacent plants. The pupal duration is
variable, being generally longer in the cooler months. It is about 42 days in
October, 32 days in November, to 18 days in December.
-
Flight Period in South Australia
-
Probably only one brood a year, with an extended flight period from mid-October
to early March, and with the bulk of the butterflies flying during late November
to early February. Males tend to emerge earlier in the season than the females.
In captivity, a summer brood can be completed in about 14 weeks, and it is
theoretically possible for two broods to occur in South Australia during a season,
although this cannot be substantiated in the field. Both early and late instar
larvae can often be found together on the one hostplant.
Two broods occur in coastal NSW.

-
Distribution
-
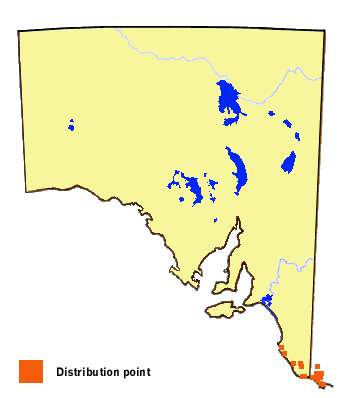
In South Australia, this butterfly only occurs in the Lower Southeast Region,
within the remant Gahnia clarkei wetlands. The butterfly has recently
been sighted in the Robe area at two small wetland remnants. Previously its
known occurrence was south of about Millicent.
The butterfly occurs as two megapopulations in Australia, consisting of a southern
megapopulation of two subspecies present in southeast South Australia, Victoria and
southeast NSW in which the butterflies possess a broad orange band on the forewings and no
yellow markings on the hindwing upperside, and a northern megapopulation of three
subspecies present in northeast NSW and southeast Qld in which the butterflies possess a
broad yellow band on the forewings and additional yellow markings on the hindwing
upperside. Where the two megapopulations meet, interbreeding produces a variable
hybrid population of butterflies. Within both megapopulations there are further
morphology clines.
The local form of the butterfly, which also occurs in western Victoria, used to be
known as subspecies antoni Tindale, but it is now included in subspecies
albifascia, which has an extended distribution to eastern Victoria and
southeastern-most NSW. The local butterfly occurs as a distinct morphological
clinal form that has more brighter and exaggerated markings than in butterflies
from populations in eastern Victoria.
-
Habitat
-
In South Australia this butterfly occurs only within the Gahnia clarkei wetlands,
which are present only within the Lower Southeast Region. These wetlands are fed by
permanent freshwater springs, and occur south of Latitude 37° 30' (approximately a line
through Millicent to Nangwarry) and also along a narrow northern coastal extension to
the Robe area.
-
Conservation Status in South Australia
-
Considered to be Rare in South Australia. Due to the historical, widespread
destruction and degradation of its wetland habitat, the remaining habitat
occurs as disjunct fragments in which the butterfly is presently known from
only five localities, where it is rarely seen. However, there has been a recent
concerted effort by local environmental authorities to preserve and fence-off
these remaining wetland fragments, and as a result the butterfly is presently
considered to be secure. The butterfly can be locally common in the eastern states
(as local clinal forms) where its preferred habitat is more extensive, although
there too its wetland habitats are being incessantly destroyed by indiscriminate
urban and small-scale agricultural expansion.
-
Threats
-
Where not protected, its Gahnia clarkei wetland habitat continues to be
sureptitiously drained, cleared and degraded. This habitat has been severely
fragmented in the past in the Lower Southeast. Draining of the wetlands puts
the habitat under stress in summer, often causing the Gahnia to tightly
inroll its leaves to the fatal detriment of the larvae of this butterfly. As
this habitat is now very limited, introduced woody weeds such as blackberry
and Coprosma have the capacity to be a serious threat by choking off
both hostplant and habitat.
-
Conservation Strategy
-
Clearance and drainage of the remaining Gahnia clarkei wetlands in the
Lower Southeast needs to totally cease, and these wetlands need to be fenced
off from grazing domestic animals. Where possible, the exotic blackberry and
Coprosma should be controlled or removed from these wetlands.
An attempt could be made to reintroduce the butterfly to some of the protected
sites where the butterfly no longer exists, and hostplant density could be
improved by the planting of new plants. The butterfly is a good survivor, and
can exist in small areas of wetland provided both are left alone.




