-
Larval Food Host
-
*Oxalis corniculata ssp
corniculata (yellow wood-sorrel),
O. perennans (native sorrel or creeping yellow oxalis) (Oxalidaceae).
The plants are perennial herbs. The larvae will eat all parts of the hostplant,
but prefer the leaves and flowers.
-
Larval Attendant Ant
-
Larvae are attended by numerous small, common black ants Iridomyrmex
sp (gracilis and rufoniger groups). The relationship is obligatory although
the early stages can survive in captivity without the presence of ants. Interestingly, the
pheromones of each ant colony that may be looking after a Small Copper colony must be
different to distal ant colonies as translocation experiments of butterfly larvae between
distal ant colonies results in the demise of the larvae.
-
Eggs
-
Large, bluish-white, sub-conical, with a very fine reticulated pattern. The reticulations
are irregular. Egg apically flattened with a depressed micropylar area. Laid in small or
large, single layered clusters of up to 20 eggs on the hostplant during spring, usually
on the underside of the leaves. In summer and autumn the cluster numbers are up to 6 eggs.
The female covers the eggs with abdominal hairs during the laying process. The latter can
be very laborious, and females will settle on or near the hostplant in the presence of ants
and remain inactive for 10-15 minutes before attempting to lay eggs. The female will only
lay in the presence of attendant ants, and if an ant chamber is not already present at the
base of the chosen plant, then the ants will quickly construct a chamber to receive the
larval hatchlings. The eggs hatch in about 13 days.
-
Larvae
-
The first instar has dark brown dorsal longitudinal lines, and there are long dorsal
and lateral setae. Upon hatching the larvae are initially gregarious for a few days
and remain on the hostplant but soon become independent and find their way to ant
chambers in the ground.
The final instar is long onisciform (slater) shape 16-18 mm long, the lateral surfaces
bearing many short bristly hairs. Head small, smooth, black, hidden beneath the body.
The colour is pale grey brown, with a pair of broad brownish-black longitudinal
dorso-lateral lines, edged white dorsally, and with a narrow dark-brown dorsal line,
and additional very narrow dark-brown lateral lines. Large shining black dorsal plates
are present anteriorly and posteriorly. The larva is covered in short dense simple
secondary setae, having slightly expanded blunt ends. Moult skins are not eaten.
Larvae feed during the cooler periods of the day accompanied by many attendant ants.
At night they rest below ground inside ant nests or specially constructed chambers
for the larvae. The ants are not normally nocturnal, but on hot nights the ants may
venture out in which case the larvae may also accompany the ants. Larvae always
return to the same ant chamber after feeding. During the hot dry summer months as
the hostplant deteriorates the larvae progressively eat the leaves, then the bark
from the stems and finally the outer membrane of the seed capsule. Eventually the
plant may completely dry out above ground (the plants are perennial). Larvae have
been observed to survive for 3-4 months over the hot summer period, scrounging and
subsisting on any new bud growth that may occur after infrequent rains, but with the
following autumn rains the larvae again become active as new shoots develop on the
hostplants. During such periods the larvae live off their own available fat supplies
and have been reported to shrink in length from 12 mm to 4 mm.
During periods of no hostplant growth, the larvae are not believed to eat the roots of
the hostplant (even though they have been seen to make digging attempts; the ground
would be very hard anyway), or be fed by the ants, or eat other larvae in the colony
(Lycaenid larvae can be cannibalistic). The larvae are believed to aestivate in the
cool underground ant chambers as best they can, awaiting any rains to promote new
growth on their hostplants. Field and captivity observations by Hunt for larvae from
the Mt Lofty Range suggest the limit of larvae survival in that area while in
aestivation mode is about seven weeks. How such colonies can survive in areas distal
to the Mt Lofty Range in the summer dry areas of its flight range elsewhere in South
Australia is not presently understood.
There are sometimes population collapses as the early stages build up to excessive
numbers that cannot be supported by the available hostplant. Hunt once counted 226
larvae on a small area of hostplant, and had to relocate many of the larvae to nearby
hostplants to prevent such a collapse. During such collapses the stressed larvae
become anaemic and the attendant ants lose interest in the larvae, sometimes even
attacking them. Paradoxically, there can be also be population explosions, with
reports of sudden mass adult emergences, that quickly disperse over a few days.
The larval period requires five instars, which can be completed in four weeks.
The extent of parasitoid wasp attack is not fully known although Ichneumonid
wasp attack has been reported by Hunt.
-
Pupae
-
Greenish or brownish cream colour, sometimes darker above, the wing cases yellowish,
smooth, shining, long cylindrical about 10-12 mm long, rounded anteriorly and
posteriorly, the abdomen is strongly arched. Pupation usually occurs within the ant
chambers and are weakly attached to the silked substrate by anal hooks. Sometimes
they are attached to the lower surfaces of debris or rocks near the hostplant.
A central girdle has not been reported. The pupae are fragile and are easily damaged.
The butterflies normally emerge from their pupae during the morning, and crawl out of
the chamber and sit on or near the entrance hole before expanding their wings,
sometimes even blocking the hole to ant traffic, but immune to ant attack. The empty
pupal shells are also eventually destroyed by the ants after the adult butterflies
emerge. Mating with females often takes place soon after wing expansion. The pupal
period is 10-13 days in spring-summer.
-
Flight Period in South Australia
-
September to early May. In hot areas the main emergence is during late spring, with
another lesser emergence in early autumn if hostplant has been available. Eggs laid
during the spring usually produce a flight either in summer or autumn depending on
the availability of hostplant. Eggs laid during autumn produce the spring flight.
In cooler moist areas the emergence is mainly during summer. If the availability of
hostplant is irregular then some larvae may take a full year to develop.
Under favourable conditions with a continuous supply of hostplant they are capable
of continuous broods over the warmer months that can be completed in about 8 weeks.
They overwinter mainly as larvae, but some pupae are believed to develop during
winter to produce the early spring flight.

-
Distribution
-
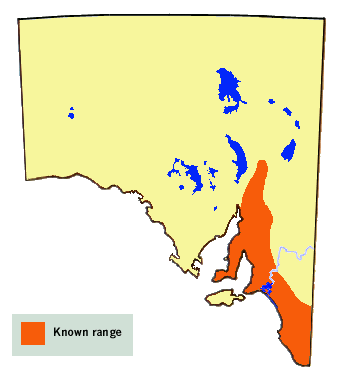
The butterfly is presently known from the Mount Lofty and Flinders Ranges, and the
Southeast Region. It has historically been recorded from Yorke Peninsula, but has
yet to be recorded from Eyre Peninsula or Kangaroo Island. Although its native
hostplant is widespread throughout the state, known butterfly colonies are
restricted to areas receiving more than about 250 mm of annual rainfall.
Based on the distribution of its larval hostplants and similar habitat areas,
the butterfly is likely to be more extensive than its present known range.
Also recorded from the higher rainfall areas of Victoria, New South Wales and
southern Queensland.
-
Habitat
-
The butterfly is usually seen in moist grassland or very open woodland areas
in which the grasses or understorey are sparse and open. The butterfly appears
to best survive where its hostplants can remain alive (at least in perennial mode)
over the hot summer months.
-
Conservation Status in South Australia
-
Rare.
-
Threats
-
Its habitat is prime agricultural and grazing land, and its existence is therefore
under constant threat from agricultural processes, particularly the effects of
herbicides, insecticides, stock trampling and overgrazing. Colonies should be able
to survive a light grass-fire, but not regular burnoffs.
-
Conservation Strategy
-
Specific grassland habitat may have to be conserved for the long-term survival of
the butterfly. Some grazing by native or domestic animals is beneficial as it
stops the grasses from becoming too rank and obliterating the hostplants.
The butterfly would be a good candidate for reintroduction into conserved areas,
as eggs or adults. Council reserves and parks that receive some watering should
be ideal.
-
Acknowledgement
-
Much of the biological information for this butterfly was provided by the late Lindsay Hunt
who was fortunate to have a colony occur on his farming property at Wistow in the Mt Lofty
Range (Victorian Entomologist 36:5 2006).







