-
Larval Food Host
-
In South Australia the larvae are known to feed on
Glinus lotoides (Hairy Carpet-weed)
and G. oppositifolius (Slender Carpet-weed) (Molluginaceae).
Other host-plants in Australia that also occur in SA include
Tribulus spp incl. *T. terrestris (Caltrop) (Zygophyllaceae).
These plants are annual or perennial herbs. Overseas, the larvae also feed on
*Amaranthus spp incl. *A. deflexus, *A. viridis (Amaranthaceae);
*Euphorbia spp (Euphorbiaceae);
flowers of small legumes incl. *Trifolium spp (clovers) (Fabaceae);
and *Oxalis corniculata (Oxalidaceae).
The larvae will eat all the soft parts of the hostplants, including leaves, flowers and fruits.
They will burrow into flower buds and fruiting capsules and eat out the contents.
On Glinus lotoides, which has a dense tomentose covering, the larvae will
eat the firm surface parts of the leaves, leaving behind the fluffy tomentose-down.
-
Larval Attendant Ant
-
Larvae are usually attended by a few small black ants, Iridomyrmex
and Monomorium species.
-
Eggs
-
Small, initially pale green, later turning greyish white, hemispherical,
strongly flattened top and bottom, slightly depressed on top with a small
darker central micropylar area. Ornamented with a coarsely reticulated
pattern on the side that becomes finer on top. The reticulation facets on
the side are trigonal, which are grouped together in an orderly fashion to
form large hexagons. The facets on the top of the egg are much smaller and
are of irregular shape but tending to hexagonal shape. There are short, thick,
blunt spines at the pattern intersections on the side of the egg, which are
absent on the top of the egg. Laid singly on the foodplant, which can be the
flower buds, leaves and other softer green parts.
-
Larvae
-
The first instar is initially pale yellow, changing colour after eating the foodplant,
with long lateral and long recurved paired dorsal hairs. The head is black.
In subsequent instars the larvae become covered in secondary setae, and the long hairs
become progressively shorter with each instar.
The mature larvae are about 10 mm long, onisciform (slater shaped) with an indistinct
longitudinal dorsal furrow. The head is small, smooth, dark brown to yellow brown,
hidden beneath the body. The body is covered in numerous, pale coloured, short hairs
that are longest dorsally and peripherally. The tiny secondary setae are similar to
the hairs having a long erect or recumbent simple pointed central spine set on a raised
stellate base having about 5-8 rays. The secondary setae impart a scabrous appearance to
the larvae. The posterior dorso-lateral organs are well developed. The larvae are
coloured some shade of green with darker coloured longitudinal dorsal and lateral lines,
and hachured subdorsal lines. The darker lines are sometimes brownish. The lateral line
is marked further with a very narrow white line. Some larvae are strongly brownish
coloured, which usually indicates parasitisation. The colours are cryptic and blend in
with the portion of the foodplant they are eating. Larvae feed openly during the day.
The presence of larvae on the foodplant is readily discernible by holes in the buds and
by the presence of small ants which usually indicate the position of actively feeding larvae.
-
Pupae
-
Short cylindrical, rounded anteriorly and posteriorly, about 8 mm long, with numerous
short bristles on the abdomen, thorax and head but which are absent ventrally and on
the wings. Subtranslucent pale green when freshly pupated, later becoming opaque.
Sometimes yellowish, and a few days before the adult butterfly emerges the pupae often
turn brownish. With variable amounts of fine black speckled markings, and with a black
dorsal line and a broader broken subdorsal line. The latter is sometimes nearly absent.
Weakly attached by anal hooks and a central girdle, either to some part of the foodplant
where it touches the ground particularly near the main stem, to leaf debris beneath the
foodplant, or within ant chambers below ground around the main stem where they may be
attached directly to the stem or the soil. The pupating larva will sometimes loosely web
together a folded leaf or two leaves, within which it will pupate. The pupal period is
variable. In the Far North of SA the pupal period is 13-16 days in mid-spring. It can
be as short as 5 days in hot tropical areas.
It is not known if the pupae have the ability to aestivate, to adjust to the seasonal
growth of the host plants. In captivity the pupae always mature quickly.
-
Flight Period in South Australia
-
The only South Australian records are for late September-early October in the Far North,
early March in the Riverland, and late April-early May in the Far Northeast. In nearby
New South Wales and Victoria it has been seen flying from January to May. There is also
a record from the Alice Springs area to the north of SA at the end of December. In SA
it is likely the butterfly would fly throughout the warmer months wherever its host plants
remain in a green, growing condition, and in this situation would be dependant on seasonal
rains or the presence of water. The butterfly is seen all year round in the tropical north
of Australia. It has not been reported what the butterfly does when the host-plants die-back
through lack of water. A brood can be completed in less than seven weeks during early spring
in the hot Far North areas of the state.

-
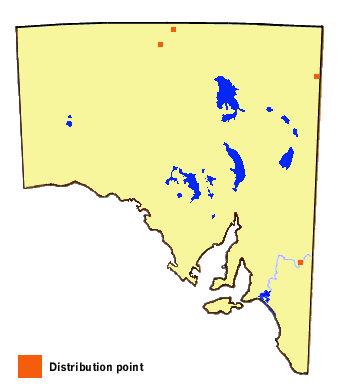
Distribution
-
This butterfly normally requires a tropical to subtropical environment but sometimes ranges
into sheltered, hot temperate areas. In South Australia the butterfly has at present been
encountered at Berri on the River Murray, and also north of Oodnadatta in the Far North,
and again near Innamincka in the Far Northeast. Its Glinus and Tribulus
hostplants have been widely recorded in the Murray-Darling Basin and also along the ephemeral
river system associated with the Lake Eyre basin in the Far North pastoral areas of South
Australia, and so there is a good chance the butterfly will be further encountered in those
regions. There are also records from the adjacent Alice Springs area to the north of SA
and also to the east in the Mildura and Lake Hattah areas of northwest Victoria, and from
Lake Victoria in southwest NSW. It occurs more commonly in the tropical north of Australia,
and in the hot, humid, open woodland areas of New South Wales and Queensland. The butterfly
also extends throughout tropical Asia and on into the northern Mediterranean, but is replaced
in Africa by a very closely related species Zizeeria knysna.
The early stages of known breeding populations of the butterfly in the Far North Region of SA
are heavily parasitised by flies and wasps, and this would suggest that these populations are
relatively sedentary. However, the butterfly probably has some degree of nomadic dispersal
ability (typical of the Polyommatini group of lycaenids), with breeding stocks being replenished
from interstate due to the butterflies following the major creek-lines down into South Australia
during periods of good inland rain.
-
Habitat
-
In the Riverland area of South Australia and the adjacent areas in New South Wales and Victoria,
the butterfly's host-plants are associated with the floodplains and irrigation areas of the
River Murray. In the Far North and Northeast of the state the Glinus (carpet-weed)
hostplants are associated with waterholes along major ephemeral creek systems. The Glinus
plants are sometimes also associated with saline, alluvial clay depressions and also have
a particular liking for high nitrogen, resulting from animal effluent concentrated in such areas
as stockyards and around waterholes and man-made dams.
-
Conservation Status in South Australia
-
Presently classed as rare in SA Its preferred Glinus hostplants have particular
growing requirements and are not common. In the Riverland the butterfly has not been seen
since the original specimens were observed in 1962, and it may be extinct in this region
of South Australia, although it was last reported from nearby Mildura in 1977.
-
Threats
-
The Glinus hostplants are associated with waterholes in the Far North and
Northeast of the state. Most of these waterholes are on pastoral leases and
consequently the plants are likely to suffer from cattle trampling. It is not
known if the cattle also eat these plants (the plant is not listed as being
poisonous), but if so then the habitat of this butterfly would likely suffer
considerably in this situation. These same waterholes would also be affected
by periodic floods and as a consequence the butterfly would have to re-establish
itself from adjacent unaffected areas, perhaps even from interstate depending on
the degree of local habitat degradation. In the Riverland the Glinus
plants grow in the floodplain environment, and since there has not been too many
floods in the Murray River system over the past 30 years due to the massive uptake
of water by irrigators, this plant is now very rare. Caltrop is a terrible burr-weed
of orchards and pastures in the Riverland and is supposed to be eradicated on sight.
-
Conservation Strategy
-
The carpet-weed (and any other native hostplant other than caltrop) needs to be
re-established in the artificially produced flood plain areas that are now being
managed by various farmers and conservationists along the River Murray system.
The same hostplants could also be grown in council reserves in the towns along
the River Murray. In the Far North and Northeast of the state, carpet-weed could
be reintroduced to favourable creeks on conserved lands, or even artificial
waterholes could be created in similar situations that would also suffice to
provide water to other flora and fauna in the region.








