Bitter-bush Blue
Theclinesthes albocincta (Waterhouse)

Interesting Aspects
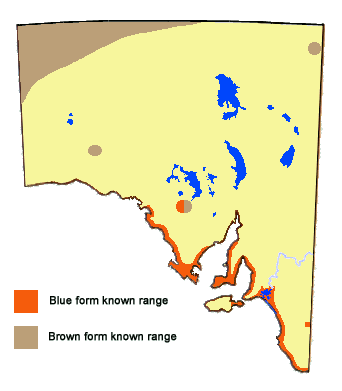
This butterfly belongs in a genus having very closely related species that are still in active phenotypic and polyphenetic processes of speciation, wherein each of the species has evolved on specific larval hosthosts in often-specialised ecological niches. It is sometimes difficult to distinguish the various adult species, especially the females as the latter usually have similarly patterned blue coloured wing uppersides. Theclinesthes albocincta has evolved on Adriana (Euphorbiaceae). Even within this species there is active speciation.There is a blue coloured adult form having larvae that exist solely on Adriana quadripartita form klotzschii, and this butterfly only occurs in coastal regions of South Australia.In the hot inland dune areas of Australia and also in northwest Western Australia, there are other forms of this butterfly that exist solely on Adriana urticoides (in its various growth forms). These butterflies are brown coloured, without any blue markings. In coastal and near coastal areas of Queensland and northeast New South Wales there are blue and green forms of the butterfly that survive on Adriana urticoides form glabrata.
In coastal southwest WA, there exists a purple coloured (male) adult butterfly, very similar in pattern to the above adults, but which exists solely on Adriana quadripartita form quadripartita. This butterfly is presently known as Theclinesthes hesperia. It was probably the original archaic species from which T. albocincta evolved. Another (but more widespread) purple coloured (male) adult butterfly within the group has evolved on Acacia (Leguminosae), and this butterfly is known as Theclinesthes miskini. Yet another purple coloured (male) adult butterfly, very similar in appearance to the previous butterfly, but which tends to be larger in size, has evolved on cycads (Cycadaceae) and macrozamia (Zamiaceae). This butterfly is found in coastal and near coastal areas of eastern and northern Australia, and is known as Theclinesthes onycha .

There are a further two species in the group that are slightly more divergent in morphology, and which have a similar speciation background as for Theclinesthes albocincta. One of these species is a blue coloured adult butterfly which has evolved on the common and widespread saltbushes (Atriplex, Rhagodia, Chenopodium etc, Chenopodiaceae) and is known as Theclinesthes serpentata. However, there is also a similar but slightly smaller butterfly without blue colouration (as for T. albocincta) which has evolved on coastal samphire (Chenopodiaceae) in eastern and northern Australia. This butterfly is known as Theclinesthes sulpitius . These various species of Theclinesthes have collectively presented Australian taxonomy experts with the unusual predicament of trying to determine, as to what stage should a geographical form of a butterfly be recognized as a subspecies, and at what stage should a subspecies be recognized as a distinct species.There is currently a DNA molecular project underway by Rod Eastwood at Griffiths University to try and resolve this dilemma. Many of the Theclinesthes forms and even the presently recognised morphological species are genetically very closely related, to the point where they would not normally be recognised as separate species. Some have only 'speciated' during the Pleistocene geological period, less than 2.6 million years ago!
Adults of T. albocincta normally fly very close to the hostplants, but males will dune-top during the heat of the day, especially when females are scarce. Males attempt to set up mini-territories on the hostplants, from where they make frequent flying forays to either fend off other males, or to other adjacent hostplant areas to look for unfertilised females.They perch on twigs or bushes in these territories, usually with opened wings. Females are normally seen flying near the hostplants where they are constantly searching for a good place to lay eggs.On occasions there are mass emergences of the butterflies when hundreds of butterflies are seen flying around the hostplants. Flight is reasonably fast, but of short duration, and occurs at the level of the hostplant bushes which are normally below (human) head height. Both sexes can usually be approached with ease when settled.
This butterfly is probably the most variable coloured butterfly to occur in Australia. The areal extent of the blue areas on the wing uppersides of the coastal form butterfly is variable, and reduces rapidly for butterflies occurring away from the coast or in hot, dry areas. The inland form of the butterfly occurring in the Far North of the state is entirely devoid of blue colouration.The underside colour can vary from pale to dark grey, or can be yellowish or brownish, and the markings can also be variable. Butterflies flying in winter are much darker on the underside (sometimes black and white) thought due to increased melanin content being acquired during the longer early stage period in a cold environment (and perhaps shorter daylight period), and in some populations the blue upperside colour is replaced by dark grey colour during winter. This dark colouration cannot be reproduced in captivity with the pupa and is thought to be acquired through the larval stage.