-
Larval Food Host
-
Capparis species including C. mitchellii (tree caper),
**C. spinosa (bush caper) (Capparaceae). In central west
New South Wales and southwest Queensland the hostplant is a broom like tree
Apophyllum anomalum (Capparaceae), which does not occur in South Australia.
There are numerous other species of Capparis growing in the tropics of Australia.
The larvae eat all the soft green parts of the hostplant, and if that has been demolished
then they will also eat the green bark off older twigs. There is also a record of
Cassia sp (Leguminosae/Caesalpinioideae) being a hostplant near Alice Springs,
but this requires further confirmation.
The bush caper,
a domesticated cosmopolitan version of caper now grown in hot areas throughout the world,
is being more commonly grown in home vegetable gardens in South Australia.
During migrations, some females will lay eggs on plants that are not recognised hostplants.
Banksia ornata and B. serrata (Proteaceae) and Correa baeuerlenii,
cultivated orange and Zieria sp (Rutaceae) have been reported to be used for this
purpose. In the case of the Correa, more than 100,000 eggs were estimated to have
been laid on a single plant in northeast Victoria (Faithfull, 2005). None of the emergent
larvae ate the leaves of this plant, and all died. The reason for this 'egg dumping' is
not known, but could be because the female is continually producing eggs within her abdomen,
and because suitable Capparis plants do not exist in temperate areas suitable for
egg laying, she may have to expel some of these eggs to stop her abdomen from becoming too
distended or heavy. Often the laid egg clusters contain infertile eggs. The active aromatic
oils in Capparis are mustard oil glycosides, which provide the egg laying stimuli
for the females. These oils are absent in Proteaceae and Rutaceae, but are notably present
in Brassicaceae, yet interestingly, the females do not utilise the latter for 'egg dumping'.
-
Eggs
-
Pale yellow when newly laid, later turning orange, spindle shaped with the height about
twice the diameter, the lower half tapering to a flat base, the upper half tapering to
a blunt point, with coarse vertical ribs numbering about 14-16 most of which continue
to the top of the egg producing a rosette pattern when viewed from above. There are also
fine horizontal ribs. Laid in large batches on the new green growth of the hostplant
including the flower buds. An individual female has been observed to lay 114 eggs within
30 minutes (an egg every 15 seconds), on both sides of a caper tree leaf, while the
collective result of several females may result in an egg batch considerably more than
this number. The egg laying capacity of individual females is not known. During a large
migration flying east through Adelaide in the late spring (17-19 November) of 1975 it
was estimated that one large cultivated tree caper received more than 40,000 eggs.
During another big historical migration through suburban Sydney, the great lepidopterist
Dr. G. A. Waterhouse estimated 250,000 eggs were laid on a similar large tree caper in
his garden. Eggs hatch in 4 days during spring in Adelaide.
-
Larvae
-
Initially pale orange, long cylindrical shaped, the skin is shiny, with dark hairs (setae)
arising from small brown coloured, simple raised bases. The hairs are longest at the
anterior and posterior ends of the larvae. The head is black, large, rounded, smooth and
shining, with a few dark hairs. The larvae eat their egg shells first, then follow with
whatever the eggs were laid on. Initially, these immature larvae feed gregariously by
scouring the leaf, bark and bud cuticle, and the orange larvae colour quickly changes to
greenish-brown after the larvae feed on the hostplant. Older larvae will devour whole
leaves and young stems.
All stages of the larvae have special glandular setae (hairs), which secrete poison,
visible at the ends of the hairs as clear coloured droplets. It is best developed in
the early instar larvae. The composition of the fluid is unknown, but is likely to
be a deterrent to both invertebrate and small vertebrate predators, as the hostplants
contain irritant glucosinolates, which the larvae are capable of assimilating and
converting into poisons. This affords some protection to the larvae while they feed
openly on the hostplant during the day. Young larvae are not bothered by the large
and fiercely predatory meat-ants, and this is probably due to the fluid droplets on
the larval hairs. Larvae start to produce the poison to the hair tips soon after they
emerge from their eggs.
Subsequent instar larvae are mostly brown coloured, and acquire increasingly abundant,
short setae, which impart a rough scabrous appearance. The bases of these hairs are
yellow, some of which develop into small protuberances. They also develop white
lateral hairs.
Mature larvae are long cylindrical shaped, about 34 mm long, the skin on the dorsal
surface is mostly a shiny brown colour with numerous small raised yellow spots and
protuberances from which arise very short hairs. Those hairs that arise from the
protuberances are slightly longer and are poison tipped. The larvae are mostly white
laterally, with white lateral and sublateral hairs which arise from simple raised
white bases. The head is black with white hairs arising from raised yellow or white
bases, and there is a prominent white inverted V shaped marking on the front.
The young larvae are gregarious, but the older larvae are more independent. They
cluster on the young growth and feed openly during the day. They are voracious
eaters, and will pupate prematurely if the food supply is inadequate, producing
adults that are undersized. The larval duration is about 3-5 weeks in Adelaide,
depending on the average day heat.
-
Pupae
-
Angular elongate typical for the subfamily, about 23 mm long, marked black and white
like bird droppings which they imitate, the amount of individual colour is variable,
and sometimes there is some pink colouration. The proportion of white colour compared
to black tends to be more common when the average day heat is high. Some pupae can be
nearly all black, while at the other extreme, some can be nearly all white. The head
of the pupa has a short blunt anterior projection, the thorax has a dorsal ridge
produced into a central obtuse point, a further short projection occurs at the wing
base, and there is a prominently pointed dorsolateral abdominal projection. There is
an obscure abdominal dorsal ridge. There are also a longitudinal series of very small,
paired yellow dorsal protuberances, a similar row of dorsolateral protuberances, and
with a few additional yellow protuberances behind the head and on the thorax. The
proboscis (on the ventral side of the pupa) extends slightly beyond the wing areas along
the abdomen, which seems to be a common attribute of the subfamily. Attached by a
cremaster and central girdle to the outer parts of the hostplant, or occasionally to
nearby vegetation. On Capparis they are usually attached one per leaf if any
leaves remain, otherwise they are attached to the bare twigs. On Apophyllum
a line of many pupae on a single stem is possible. The larvae make sure the caper leaf
remains attached to the tree for the duration of the pupal period by spinning silk
between the base of the leaf and the stem. The pupation position is random, and pupation
may occur with the head pointing either upwards or downwards, although the former
position seems to be preferred. The pupal period is about a week during the warmer months,
increasing to two weeks during the cooler months.
The butterfly numbers are kept in reasonable check by predators and parasitoids during
the entire life history, although the efficiency of this check must fluctuate giving rise
to the periodic immense numbers of migratory butterflies. When Dr G. A. Waterhouse
("What Butterfly Is That? " Angus & Robertson 1932) made his investigation on the life
history of the butterfly when a large migration of butterflies passed through Sydney and
laid eggs on a large cultivated tree caper in his suburban garden, he estimated 250,000
eggs were laid on his tree by the females. The eggs were immediately set upon by egg
sucking invertebrates. Hatching larvae were similarly dispatched. Surviving larvae were
systematically removed by wasps to feed their young, and finally the larvae were set upon
by parasitoid flies. There were still sufficient larvae to totally demolish all the edible
green parts of the tree. Some pupae managed to survive the parasitoids and produce
butterflies which were then set upon by birds. Waterhouse estimated that less than 50
butterflies actually survived to fly away from his garden!
-
Flight Period in South Australia
-
In the Far North pastoral areas of the state it is possible to find odd butterflies
throughout the year. In the southern settled areas it is most commonly seen in the
spring and early summer during its main southerly migration. It is sometimes seen
again in fewer numbers in these southern areas during late summer or autumn, usually
due as a result of a secondary migration from the northern breeding areas after
summer thunderstorms or good monsoon rains have occurred in those areas. Migration
numbers reaching southern areas during spring fluctuate year to year, from common
to scarce. The brood period is about 6 weeks in southern areas, but is considerably
shorter in the hotter northern Flinders Ranges.
With the increased use of domesticated bush caper in gardens in South Australia the
butterfly will sometimes maintain a presence around those bushes if permitted. It
has recently been reported to overwinter as pupae in warm sheltered, maritime areas
on Kangaroo Island where the bush caper is being cultivated, with all life stages
of the butterfly apparently being observed during August. Further recent observations
however, suggest it cannot survive the winter in the northern Flinders Ranges on its
natural hostplants.

-
Distribution
-
Due to its migrant and vagrant habits, the butterfly can occur throughout mainland
Australia, with the odd occurrence on Tasmania. It regularly makes the crossing to
Kangaroo Island during late spring over the 20 km expanse of seawater in the
Backstairs Passage that separates the mainland Fleurieu Peninsula from the island.
Its hostplants normally grow in tropical and sub-tropical latitude areas, and do not
occur in temperate areas except where they have been carefully cultivated. The
butterfly is more commonly observed in South Australia during its migratory phase.
It will take up residence in southern settled areas over the warmer months if its
hostplants are present, but the butterfly cannot normally survive the winter in
these areas.
It is a very strong migrant, and at irregular intervals immense numbers fly south from
the tropical and subtropical latitudes, reaching as far south as Tasmania. Since
European settlement, descriptions of such flights have appeared periodically in newspapers
and natural history publications. Unfortunately, these migratory flights are not
very well documented scientifically. It is not known whether they occur as a single
immense migration or occur in fits and starts as butterfly numbers build up at any point
along the migratory path. On their flight southwards into South Australia they are
helped along by the strong easterly and northerly winds prevalent during the late spring
and early summer. Local observations indicate that after moving southwards, they
turn around and return northwards again to their breeding grounds, a minimum round trip of
1300 km through Adelaide. Numbers in the migrations are sometimes immense, and their
bivouacs are unforgettable to those who are lucky enough to see them, reported as
'butterfly snow'. In these situations the butterflies totally cover every bush in
the area, often with butterflies hanging six deep off each other.
During the recent big migrations in South Australia (1999)
(2005) that started somewhere in the northern inland regions of Australia, the migrations proceeded
southerly to the Lower Southeast Region of South Australia and to southwest Victoria, then turned
west to the Fleurieu Peninsula, Kangaroo Island and Yorke Peninsula, and then made a northerly turn
before disappearing back north, presumably from where they started. The 1999 migration passed
through Adelaide over two days (30 November 1999 - 1 December 1999), during which time individuals
were moving N-NNE into a northeast wind. It was noted on the first day that some 650 individuals
passed over a 50 m line of sight in one hour during a peak period of movement. This sighting was
at the edge of the Adelaide Plains of suburban Adelaide along the foothills of the Mt Lofty Range,
which probably helped funnel the main migration front. On the second day the peak flight numbers
had reduced to about 440 individuals per hour over a 100 m line of sight. On 2 December the
migration had essentially finished in Adelaide. Butterflies were also recorded flying earlier
through Port Germein (23 km north of Port Pirie) on 27 and 28 November where they were migrating
north-easterly in peak numbers of 474 individuals per hour in front of the person making the
observation. It is not known if this mass had taken a short cut or were part of the main migration
that had made an earlier circuit through Yorke and/or Eyre Peninsulas that bypassed Adelaide.
Ian Faithfull documented a massive migration near Armidale in northeast NSW with a front of at least
one kilometre in width over the period 21-27 November 1975. The daily flight occurred between 9 am
and 8 pm, and was generally N to NNE with numbers peaking on 23 and 24 November and estimated at
10 butterflies per metre per minute. Most flew about head height but some flew very high in the air
estimated at 100 m. It is not known if this migration was the same, or contained butterflies from
another major migration that passed through Adelaide on 17-19 November 1975 and flying in an eastwards
direction!
-
Habitat
-
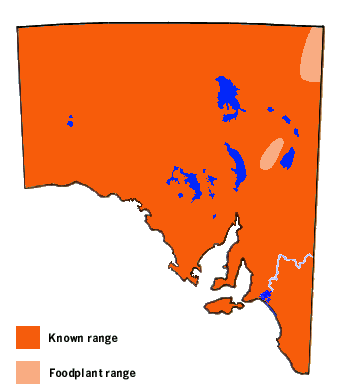
The Capparis hostplants are extremely common in the northern half
of Australia, and can be found in most habitats. Within South Australia
its hostplants have only been recorded to grow naturally in the northern
Flinders Ranges and in the extreme northeast area of the state. The
hostplant in these locations is the tree caper, which occurs sparsely
and is unlikely to produce the immense numbers of butterflies seen during
the big migrations. In the northern Flinders Ranges the plant tends to
frequent the ravines and rocky areas. The butterfly and its early stages
are not biologically suited to the winter conditions of southern pastoral
and temperate areas of South Australia.
-
Conservation Status in South Australia
-
A migrant, locally common in breeding areas and during southern migrations.
In the northern breeding areas of South Australia, numbers of the butterfly
tend to be more common about their hostplants during the warmer months.
Most of the migrating butterflies seen in South Australia probably have their
origin from the inland areas of New South Wales, Northern Territory and Queensland.
-
Threats
-
There are no major threats to the butterflies within South Australia.
In western NSW and southwest Queensland the butterfly breeds in the
plague locust belt and it is likely that the locust controls have an
influence on butterfly numbers.
-
Conservation Strategy
-
The tree caper, the only larval hostplant of the butterfly to occur naturally
in South Australia, does not appear to be reproducing itself satisfactorily
in SA due to the effects of past droughts and the grazing by animals.
An attempt should therefore be made to revegetate the tree in suitable areas.
It will require protection from both grazing animals and from the caterpillars
of this butterfly while still in the immature thorny stage of growth.








